following the fast advancement of vaccines, the FDA has gotten a number free from COVID-19 achievements for crisis use as a feature of the continuous battle against staggering infection. Up until now, we’ve seen the organization support clinical advances including lab-made monoclonal antibodies for moderate diseases that danger turning more extreme, a quick test that utilizes CRISPR quality altering tech, and Fitbit’s Flow ventilator.
The furthest down-the-line instrument to acquire leeway is the principal AI-based screening gadget intended to pinpoint sneaking indications of COVID-19 in asymptomatic individuals.
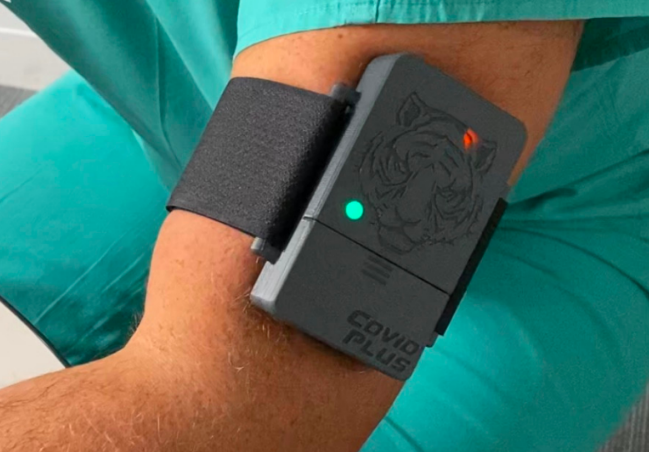
Named the Tiger Tech COVID Plus Monitor, the mechanical assembly is an armband that utilizes light sensors and a little PC processor to check for biomarkers of the infection, like hypercoagulation — a typical COVID-19 irregularity that makes the blood coagulation all the more without any problem. Once tied to an individual’s arm, the screen’s locally available sensors begin gathering beat signals from the bloodstream over a time of three to five minutes. The processor at that point extricates the vital information from the estimations and feeds them through the AI model. End-product, including whether the test shows positive biomarkers (like hypercoagulation) or is uncertain, are addressed by various hued lights.
In its declaration, the FDA features that the armband isn’t intended to analyze COVID-19 and in this manner ought not to go about as a substitute for a customary test. Nor is it intended for use on indicative individuals. All things considered, it’s expected as to a greater degree a back-up or safety measure to be conveyed close by a temperature check if the perusing doesn’t show a fever in possibly asymptomatic people.
Together, the two screening devices could be utilized to check the spread of the infection in the scope of public settings including medical services offices, schools, work environments, amusement parks, arenas, and air terminals, the office said.
The screen’s leeway follows clinical investigations in medical clinics and schools that showed comparative outcomes. In the medical clinic setting, the gadget effectively spotted COVID-19 biomarkers at a pace of 98.6 percent, while additionally precisely recognizing those that didn’t have the indications at a pace of 94.5 percent. The school preliminary basically approved the outcomes, as per the FDA.
Also Read >>>> Google’s Pixel Buds drop to $159 at B&H Photo and Best Buy